Professor Penny Vlahos investigates what happens with the ocean chemistry at the marginal ice zones in her recent publication
By Ewaldo Leitao.
The Arctic Ocean is undergoing rapid changes due to climate change. Increasing temperatures result in decreasing sea-ice extent, constant decreasing and thinning of permanent sea-ice caps. Some projections even show a completely ice-free Summer by 2050!
Another consequence of climate change is ocean acidification due to increasing atmospheric CO2. That leads to the decrease in water pH and changes in carbon chemistry dynamics. The Arctic may be a small ocean (3% of total oceans area) but it has an important contribution to carbon uptake (10%). Therefore, it is necessary to understand the impact of these changes across the oceans, including the Arctic, in order to be prepared for it.
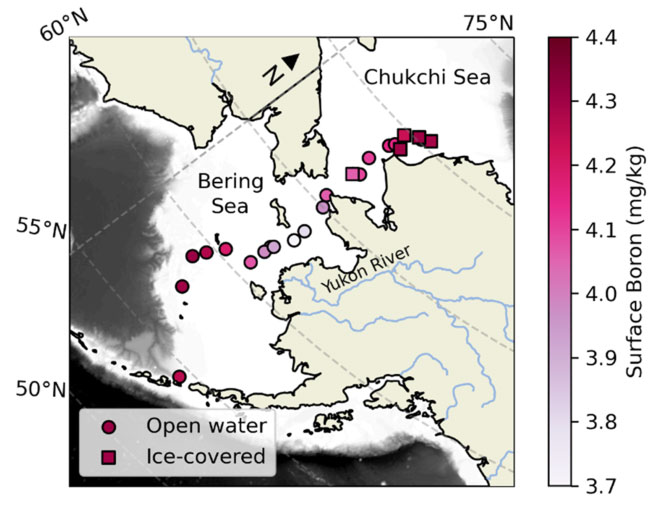
Some chemical elements, such as boron, contribute to the ocean’s capacity to resist changes in pH, that is ocean’s alkalinity. Boron, in combination with salinity, has been used as a universal rule in the open ocean (boron to salinity ratio) in order to understand the contribution of boron to alkalinity, and therefore ocean carbon chemistry. But how does that change in the less saline areas, such as the marginal ice zones of the Arctic?
In the recent paper published in Nature Communications, Prof. Penny Vlahos and graduate student Lauren Barrett observed that, when measured in low salinity areas (marginal ice zones), the boron to salinity ratio deviates from the expected in open oceans. In a cruise that took place in May of 2021 (you can read more about the cruise here), researchers were surprised to find significant deviations in the boron to salinity ratios in ice and brine samples. Lower water temperature and lower salinity alters the exchange between boric acid and borate, which is used to determine the contribution of boron to sea water alkalinity (capacity of water to resist changes in pH and acidification), driving this deviation of the boron to salinity ratio compared to open ocean waters.
The unique microenvironment of the marginal ice zones creates a very dynamic system. As seawater freezes, salts are rejected, but there is still a liquid region between ice crystals, called brine channels. These channels allow boron to undergo inorganic changes that may result in the variations observed in some of the samples, increasing the variability of boron to salinity ratio observed in these Arctic areas.
Prior to boarding the research vessel, researchers had to quarantine for two weeks. But this was a valuable time to Lauren Barrett. “Over quarantine I spent a lot of time reading about the various uncertainties that other authors encountered in accurately and precisely constraining the carbonate system in this highly heterogeneous environment. The boron to salinity ratios that we present here warn against applying universal ratios constrained in the open ocean to marginal ice zones and ice environments.” says Lauren.
Lauren also shared a little bit about her experience: “I am very grateful for the opportunity to work with our international coauthors. The collaborative and interdisciplinary nature of marine science is one of my favorite aspects of working in this field. This research cruise was a great experience both personally and professionally, and I continue to be grateful to work in a field where cruising and getting to see polar bears is all in a day's work.”
The Arctic is an important sink of carbon and yet highly susceptible to climate change. Therefore, understanding detailed information of this system, instead of applying universal ratios, is necessary in order to better understand the carbon chemistry of the Arctic and be prepared for the consequences of climate change.
Vlahos, P., Lee, K., Lee, CH., Barrett, L, and Juranek, L. (2022) Non-conservative nature of boron in Arctic marginal ice zones. Nature Communications Earth & Environment 3, 214